Artlabeling Activity Implantation of the Blastocyst in the Uterine Lining
Introduction
Receptive endometrium is an accented prerequisite for a successful embryo implantation, being defined by a limited time-frame when the endometrium is favorable for embryo adhesion and the subsequent attachment and invasion processes (1).
Endometrial receptivity is a circuitous procedure that is orchestrated by the synergistic deportment of chief reproductive hormones estrogen and progesterone, too as plead of other endocrine, paracrine and autocrine factors (two, 3). Dumb endometrial receptivity is thought to be ane of the major reasons for embryo implantation failure (4). In assisted reproductive technologies (ART), where the good quality embryos are transferred equally a standard of intendance, implantation failure remains an unsolved obstacle (5, 6). Regardless of the advances in assisted reproduction, particularly regarding the more effective ways of embryo selection and cryopreservation, many patients repeatedly fail the treatment procedure. What we are facing today is that implantation failure in Fine art is common, and we lack the evidence-based therapeutic solutions for treating it. As a effect, clinicians often experience obliged to offer treatments that are largely empirical, based on some biologic rationale just with niggling clinical evidence to support their use (7, eight). The treatment failure is every bit frustrating to both patients and their providers, which even more than emphasizes the urgent need for novel effective handling to prevent yet another failure.
The role of growth hormone (GH) in female reproduction has gained renewed interest and has get a heated topic over the concluding decade. The local GH production in the reproductive tissues themselves exert an of import autocrine/intracrine furnishings on those tissues, in addition to the pituitary product of GH (9). Moreover, local insulin growth cistron 1 (IGF-1) production (known downstream mediator of GH) has been shown to exist controlled by gonadotropins and estradiol also (10). Prove emerging from clinical practice suggests that GH administration during ovarian stimulation may improve oocyte quality [higher number of oocytes nerveless, higher fertilization rate, and college number of embryos reaching the transfer phase (11–fifteen)], increase pregnancy rate (sixteen–24), implantation rate (sixteen, twenty–23, 25, 26), and alive nascency charge per unit (12, 16, 19, twenty, 23, 25, 27). The accumulating beneficial effects of GH on assisted reproduction outcomes practise non exclude the possibility that this consequence is due, at to the lowest degree in part, to an action of GH on endometrial receptivity.
Growth Hormone in the Endometrium
GH is a peptide hormone secreted by the anterior pituitary gland, having important role in prison cell growth and metabolism throughout the trunk. GH together with its receptor, GHR, and related growth factors including IGF-ane, is expressed in the endometrium of rats and human (28–31). The report by Sbracia et al. obtained biopsies from women in proliferative and secretory phases, as well every bit first trimester decidua (from elective pregnancy terminations) (28). They showed that there was no GH expression in proliferative glandular epithelium, just GH immunoreactivity appeared in the mid-luteal secretory stage (no subdivision within secretory phase was done) and increased in the decidua from the kickoff trimester abortions, with similar expression in the decidual samples from the term pregnancies, suggesting a part in embryo implantation process. Interestingly, no stromal expression of GH was observed in any sample (28). Moreover, the authors analyzed GH expression in the endometrium from women with "luteal phase defect," defined by low progesterone levels <eight ng/mL and delayed endometrial maturation, and saw significantly lower expression of GH (28). This data suggested close relationship between GH expression in endometrium and progesterone level/function. Farther, a recent study on homo endometrial cell line indicated that GH may act in a direct or IGF-1-mediated manner on human endometrial cells to promote proliferation and vascularization and up-regulation of receptivity-related genes such as vascular endothelial growth gene (VEGF) and integrin beta 3 (ITGB3) (21). VEGF is an of import histrion in angiogenesis (32), and it has been shown to human activity in an autocrine way on endometrial epithelial cell adhesion as a primal regulator in the implantation process (33). ITGB3 is a well-known biomarker of receptivity (34), and downwardly-regulation of this biomarker (miracle detected in women with unexplained infertility, endometriosis, and luteal phase deficiency) has been related to lower pregnancy rates (35, 36).
Apart from the furnishings of circulating GH and locally produced GH on endometrium, there is a proposed indirect event of ovarian GH on endometrial part, namely its involvement in the function and maintenance of the corpus luteum (37, 38). While the majority of the information come from various animal models, they are nevertheless significant. Luteal function and its maintenance are vital for the institution of pregnancy and its viability due to the production of progesterone past the corpus luteum—the main "keeper" of the normal early pregnancy. Hence, the stimulatory effect of GH on ovarian steroidogenic jail cell function may play a major function in endometrial office and dysfunction via its effect on ovary (see Figure 1 for the proposed mechanisms of GH activeness on endometrium).
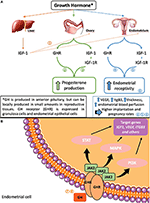
Figure 1. Possible mechanisms of GH effects on ovarian and endometrial function (A) and on endometrial cells (B). Numbers in the effigy signal studies where the information is presented in particular: 1 (39); 2 (xl, 41); 3 (42); four (23); v (21); six (43); 7 (44); 8 (45).
Clinical Apply of GH and Issue on the Endometrium
Initial reports on the utilise of GH in clinical practise come from cases of hypogonadotropic hypogonadism or panhypopituitarism (46). Later, the utilize of GH has been expanded on different patient population, such as women with poor ovarian reserve, poor responders, or with poor oocyte quality due to advanced maternal age (25, 47, 48). In general, GH assistants in the infertility clinic setting has focused on GH effects on oocyte, and the effect on endometrium has been largely overlooked.
Afterward, the attention has been turned onto the endometrium, and interesting observations accept been made suggesting positive effect of growth hormone treatment on endometrial thickness and implantation potential (see Table 1 for the studies). A case report of a patient with panhypopituitarism demonstrated improved endometrial thickness and successful implantation and pregnancy after adding growth hormone to the handling protocol post-obit multiple failed in vitro fertilization (IVF)/embryo transfer cycles (55). Alternatively, a written report on 20 patients with documented GH deficiency reported improved embryo quality, but no improvement in endometrial thickness, when supplemented with GH in IVF cycle (xv) (Table one). Below nosotros volition discuss the available literature on the utilize of GH in various clinical IVF settings.
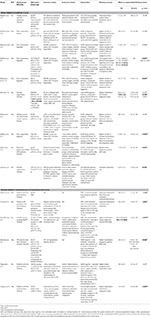
Table 1. Studies assessing the result of growth hormone (GH) co-treatment in in vitro fertilization (fresh treatment cycles and frozen embryo transfer cycles) on endometrium.
Infertile Patients With Recurrent Implantation Failure
This is a grouping of patients that fail to attain pregnancy in fresh or frozen embryo transfer cycles despite appropriate endometrial evolution (thickness and blueprint) and adept quality embryo transferred. Patients with recurrent implantation failure (RIF), having undergone three or more embryo transfer cycles after IVF treatment without a clinical pregnancy, are amidst the nigh difficult patients to treat, with no proven standard handling. Dumb endometrial maturation is suggested every bit a common cause for RIF (56–58), making it a target patient grouping who could potentially do good from GH co-assistants during IVF procedure. Chen et al. report on 42 RIF patients undergoing IVF treatment found that GH treatment throughout the stimulation increased the endometrial thickness and consequent pregnancy and live nativity rates amid immature patients <35 years old supplemented with GH when compared to no GH RIF group (19). Patients in both groups had similar summit estradiol levels and like number of oocytes retrieved (19). While it is unclear if the difference in endometrial thickness of 11.61 ± 2.ix vs. ix.7 ± ane.46 mm betwixt study and control groups, respectively was crucial in achieving higher pregnancy rates in the written report grouping, the observation is nevertheless important. This has been reported again in the second study, a randomized clinical trial including seventy RIF patients in oocyte donation program (as an ideal model for assessing GH consequence on patient's endometrium without confounding factors of ovarian historic period and response) (twenty). In that report patients, who were treated with GH throughout medicated frozen embryo transfer wheel demonstrated significantly thicker endometrium, 9.iii ± i.5 mm vs. 8.6 ± i.0 mm, respectively, and higher pregnancy and alive nativity rates compared with RIF patients in the placebo grouping (twenty) (Table one).
These are the outset two studies assessing GH furnishings on endometrium in RIF patients, and, although the findings are promising, clearly more than studies on larger patient population, as well every bit randomized clinical trials (RCTs), are needed for whatsoever clinically meaningful conclusions. It is well-accepted that endometrial thickness does not necessarily mean that the endometrium is receptive, yet it is considered as a mensurate of endometrial maturity, and optimal growth of the endometrium (>7 mm) is required for a successful embryo implantation (59–61).
Thin Endometrium
Infertile women with thin endometrium represent another potential patient population that could do good from the GH assistants. All studies on GH co-handling during treatment of infertile women with thin endometrium were conducted in frozen embryo transfer (FET) cycles, where GH was administered during the endometrial training for FET (21, 22, 43, 54) (Table 1). The largest study by Yang et al. was conducted on 225 infertile women, and did not detect whatever pregnant GH effect on endometrial thickness, while reporting significantly higher clinical pregnancy and implantation rates (22). They assessed GH effect on endometrial thickness on the day of progesterone administration, which could explicate the difference in their results from the remainder of the studies. The other three studies all noted meaning comeback in endometrial thickness on the day of embryo transfer among patients with sparse endometrium later administering GH throughout the FET cycle (21, 43, 54), and significantly college implantation and clinical pregnancy rates (21). Wu et al. written report also detected improved endometrial claret menses in the GH-administered patient group (43), like to later findings by Xue-Mei et al. study (23), who showed increased VEGF expression and improved perfusion of the uterine arteries in the group of infertile women treated with GH. In line with higher up, Cui et al. study detected VEGF up-regulation together with ITGB3 and IGF-one in endometrial cells when exposed to GH (21). The state of loftier blood menses resistance and VEGF down-regulation with inadequate epithelial growth and vascularization take been described as pathophysiologic characteristics of sparse endometrium (62), and subendometrial blood flow on the twenty-four hours of embryo transfer is related to the implantation and pregnancy rate in IVF (63). Cui et al. concluded that upwards-regulated VEGF in their study setting, in the GH group, partly resulted in the increase of subendometrial claret flow and thereby improved endometrial receptivity (21). Yet, the verbal mechanisms of GH actions on the endometrium and endometrial receptivity in general are to exist unraveled in futurity studies. Also new studies with larger written report groups and well-designed RCTs are required in order to analyze whether infertile women with thin endometrium benefit from the GH treatment.
Poor Responders
Women with poor ovarian response in Art is another patient group where GH co-treatment in stimulation protocols have been studied. All these studies (come across Table 1) take been RCTs, however with limited sample sizes, and all have reported beneficial effect of GH administration on the number and quality of oocytes and on the number of embryos obtained. Remarkably, while some improvement of endometrial thickness has been noted, those studies failed to testify whatever beneficial issue on clinical pregnancy and live birth rates (13, 49–53). Based on these findings, one could conclude that GH co-treatment in poor responders with normal endometrium does non seem to have whatever significant impact on endometrial receptivity and hence pregnancy rates. Nevertheless, nosotros should exist cautious in drawing preliminary and potentially incorrect conclusions in this type of studies without taking into careful consideration all potential confounders, including quality and number of embryos transferred, cleavage vs. blastocyst stage embryos and even type of luteal back up provided in fresh and/or frozen embryo transfer cycles (64). In addition, the full productivity rate from a single oocyte retrieval is highest when more and better quality embryos are obtained, which tin be exactly the example with GH-supplemented cycles in poor responders, resulting in higher cumulative pregnancy rates rather than per cycle success in this group of patients. Clearly, carefully designed large studies with transfers of unmarried skilful quality embryo (fresh and frozen) are warranted, albeit quite challenging to perform, in guild to analyze whether endometrial receptivity in infertile women with poor response in ART would benefit from GH assistants.
Normal Responders
Thus far, the largest group of infertile patients involved in studies on GH administration during IVF has been the normal responders (Table 1). The commencement report was performed on 240 infertile women undergoing FET, where 2 different GH supplementation protocols were compared—GH administration throughout the FET, and a single GH injection on mean solar day 8 of estrogen treatment (23). Notably, significant endometrial thickness improvement together with higher embryo implantation, clinical pregnancy, and live birth rates were detected among women with longer GH assistants (23). The authors besides noted that the longer GH improver to the treatment protocol increased the levels of estradiol, IGF-i, and VEGF serum levels, and improved perfusion of the uterine endometrial arcuate artery (23). The pulsatility alphabetize, resistance index, and peak systolic velocity/end diastolic velocity of the uterine arcuate arteries correspond the resistance of blood flow from the point of measurement downstream; increased impedance of these arteries might correlate with poor endometrial receptivity and clinical outcomes (65).
The side by side studies analyzed one,114 (sixteen) and 1,562 (24) infertile women, respectively undergoing ovarian stimulation for IVF with GH co-administration throughout the stimulation, and a positive GH effect on endometrial thickness in add-on to the higher clinical pregnancy rates was detected in study compared to control groups. GH upshot on endometrial thickness was significantly increased among older infertile women of ≥35 years onetime compared to <35 years quondam, while both groups exhibited higher implantation and clinical pregnancy rates, most likely attributed to the higher number of loftier quality embryos obtained in GH-treated groups (16). In humans, changes in GH secretion could exist historic period-related, as post-adolescence the secretion of GH decreases with historic period, which is why GH hyposecretion is observed in older patients (66). GH insufficiency can disrupt ovarian office and lead to reproductive difficulties (66). As mentioned above, in Du et al. study (xvi), GH-treated older women (≥35 years former) had implantation and clinical pregnancy rates more than two times college than those observed during IVF cycles without GH. This consequence suggested that adding GH might be beneficial for older patients.
To conclude, research on the effects of GH co-handling in Art among normal responders has been performed on sufficiently powered studies in terms of the sample size, nevertheless every bit all these studies were non randomized controlled trials, further well-designed research is needed to considerately assess the GH effect on Art outcomes in (young) women with normal ovarian reserve and normal response to ovarian stimulation.
Future Perspectives
Farther studies are warranted in order to decide the optimal dose, time, and duration of GH administration and to investigate the long-term safety of GH for patients and their offspring. The dosage and handling duration of GH differed amid conducted studies (see Table i). Because of the limited experience with the GH co-treatment protocols, at that place is a lack of evidence to back up the superiority of i over the other. In all the protocols used (run into Tabular array 1), GH was administered via subcutaneous injections, except for one study where GH intrauterine perfusion in 5 patients with non-responsive sparse endometrium was successfully used (54).
Another crucial role is to define the appropriate patient population that would truly benefit from GH treatment for improving their uterine lining quality in terms of thickness and/or receptivity. GH seems to promote endometrial growth, and its utilize could be considered in women whose endometrium does not grow and/or mature sufficiently with standard handling protocols. In addition, the electric current review concludes that even normal responders could potentially benefit from the GH administration in IVF programs, however, the improved pregnancy rates in some of the studies utilizing fresh IVF cycles could not be separated from improved embryo quality. While endometrial thickness and pattern upon GH administration has been recorded and reported, evaluation of endometrial receptivity is non as simple. Futurity studies need to focus on the molecular level in gild to evaluate the endometrial transcriptome/proteome/secretome (67), with emphasis on receptivity markers to understand and analyze the possible mechanisms of GH on endometrial receptivity. An ideal setting would exist to design an RCT with GH-supplemented mock cycles vs. command, during which endometrial receptivity could be studied on molecular level in detail (transcriptomics and/or use of commercially available endometrial receptivity tests; epigenomics and/or proteomics analyses). The mock cycle could be followed by a "true" FET cycle to enable evaluation and correlation to pregnancy rates. To sum up, undoubtedly more research on larger cohorts with carefully designed studies [every bit highlighted in a recent annotate (64)] is needed to place the patient group in whom the improver of GH to the treatment protocol in IVF programs will be nearly valuable.
Sample size and considerately designed studies (randomized clinical trials) is a delicate topic in ART as strict double-bullheaded, placebo-controlled, RCTs are hard to accomplish (68). Information technology is extremely hard to perform fully blinded RCTs in IVF because of the patient recruitment issues, where crumbling women prefer not to participate in the placebo group that requires commitment for several months of their reproductive lifespan and which ultimately may not help them achieve pregnancy (68). Understandingly, patients tend to opt for any additional treatment, toll permitting, that would potentially aid them to get meaning. Equally a upshot, the studies of GH treatment effects on IVF outcomes are rather limited on its sample size and/or are retrospective or observational in nature; nonetheless, they provide important data concerning therapeutic interventions in IVF and open upward future possibilities for improving infertility treatment protocols.
Conclusions
The electric current review summarizes the contempo data on GH co-treatment effects on endometrial parameters in assisted reproduction and proposes possible mechanisms of GH actions in the endometrium. Studies are indicating that co-handling with GH could meliorate the endometrial thickness, and perchance receptivity amid infertile women. This effect might occur through increasing endometrial blood perfusion and the expression of genes and proteins related to endometrial receptivity such equally VEGF and ITGB3 together with IGF-1, however the exact mechanisms in the endometrium remain to exist antiseptic.
Whether GH administration during IVF is useful and which patient groups could do good from information technology needs farther investigation, but the preliminary data suggest that women suffering RIF, patients with thin endometrium and older normo-responders could benefit from GH handling when undergoing ART. However, carefully designed and sufficiently powered cohort studies, RCTs, are required in the field in guild to establish the most suitable therapeutic regimen for these patients and to clarify the confusion arisen from various studies that have shown either inconsistent or conflicting findings, used small patient cohorts and/or have been poorly designed with no blinding or placebo controls.
Author Contributions
SA and LA equally contributed to the review idea and manuscript writing.
Funding
This report was supported by the University of Granada Program Propio de Investigación 2016–Excellence actions: Unit of Excellence on Exercise and Health (UCEES)–and Plan Propio de Investigación 2018–Programa Contratos-Puente, and the Junta de Andalucía, Consejería de Conocimiento, Investigación y Universidades, European Regional Evolution Funds (ref. SOMM17/6107/UGR); and the Spanish Ministry of Economy, Industry and Competitiveness (MINECO), and European Regional Evolution Fund (FEDER): grants RYC-2016-21199 and ENDORE SAF2017-87526.
Conflict of Interest
The authors declare that the research was conducted in the absenteeism of whatsoever commercial or fiscal relationships that could exist construed equally a potential conflict of interest.
Acknowledgments
Nosotros give thanks Dr. Alberto Sola-Leyva for his help in the effigy grooming.
References
two. Carson DD, Lagow Due east, Thathiah A, Al-Shami R, Farach-Carson MC, Vernon M, et al. Changes in gene expression during the early to mid-luteal (receptive phase) transition in human endometrium detected by loftier-density microarray screening. Mol Hum Reprod. (2002) 8:871–nine. doi: x.1093/molehr/viii.nine.871
PubMed Abstract | CrossRef Full Text | Google Scholar
iii. Altmäe Southward, Reimand JR, Hovatta O, Zhang P, Kere J, Laisk T, et al. Enquiry resource: interactome of human being embryo implantation: identification of factor expression pathways, regulation, and integrated regulatory networks. Mol Endocrinol. (2012) 26:203–17. doi: 10.1210/me.2011-1196
PubMed Abstruse | CrossRef Total Text | Google Scholar
6. Margalioth EJ, Ben-Chetrit A, Gal M, Eldar-Geva T. Investigation and handling of repeated implantation failure following IVF-ET. Hum Reprod. (2006) 21:3036–43. doi: 10.1093/humrep/del305
PubMed Abstract | CrossRef Total Text | Google Scholar
10. Cailleau J, Vermeire S, Verhoeven G. Independent command of the production of insulin-like growth cistron I and its bounden poly peptide by cultured testicular cells. Mol Cell Endocrinol. (1990) 69:79–89. doi: 10.1016/0303-7207(90)90091-L
PubMed Abstract | CrossRef Full Text | Google Scholar
xi. Kolibianakis EM, Venetis CA, Diedrich K, Tarlatzis BC, Griesinger G. Addition of growth hormone to gonadotrophins in ovarian stimulation of poor responders treated by in-vitro fertilization: a systematic review and meta-analysis. Hum Reprod Update. (2009) 15:613–22. doi: ten.1093/humupd/dmp026
PubMed Abstract | CrossRef Total Text | Google Scholar
12. Li X-LL, Wang L-PP, Lv F, Huang Ten-MM, Wang L-PP, Pan Y, et al. The influence of different growth hormone add-on protocols to poor ovarian responders on clinical outcomes in controlled ovary stimulation cycles: a systematic review and meta-assay. Medicine. (2017) 96:e6443. doi: ten.1097/MD.0000000000006443
PubMed Abstract | CrossRef Full Text | Google Scholar
13. Bassiouny YA, Dakhly DMR, Bayoumi YA, Hashish NM. Does the addition of growth hormone to the in vitro fertilization/intracytoplasmic sperm injection antagonist protocol improve outcomes in poor responders? A randomized, controlled trial. Fertil Steril. (2016) 105:697–702. doi: 10.1016/j.fertnstert.2015.11.026
PubMed Abstract | CrossRef Full Text | Google Scholar
14. Kucuk T, Kozinoglu H, Kaba A. Growth hormone co-treatment within a GnRH agonist long protocol in patients with poor ovarian response: a prospective, randomized, clinical trial. J Assist Reprod Genet. (2008) 25:123–7. doi: x.1007/s10815-008-9212-7
PubMed Abstract | CrossRef Full Text | Google Scholar
fifteen. Rajesh H, Yong YY, Zhu M, Chia D, Yu SL. Growth hormone deficiency and supplementation at in-vitro fertilisation. Singapore Med J. (2007) 48:514–518.
PubMed Abstract | Google Scholar
16. Du FX, Yang XH, Li J, Hao M, Goa YH. Growth hormone co-handling within a GnRH agonist long protocol improves implantation and pregnancy rates in patients undergoing IVF-ET. Arch Gynecol Obstet. (2016) 294:877–83. doi: 10.1007/s00404-016-4163-1
PubMed Abstract | CrossRef Total Text | Google Scholar
17. Hazout A, Junca Am, Ménézo Y, Demouzon J, Cohen-Bacrie P. Issue of growth hormone on oocyte competence in patients with multiple IVF failures. Reprod Biomed Online. (2009) 18:664–70. doi: 10.1016/S1472-6483(10)60011-9
PubMed Abstract | CrossRef Total Text | Google Scholar
xviii. Lattes Thou, Brassesco M, Gomez G, Checa MA. Depression-dose growth hormone supplementation increases clinical pregnancy rate in poor responders undergoing in vitro fertilisation. Gynecol Endocrinol. (2015) 31:565–eight. doi: 10.3109/09513590.2015.1025378
PubMed Abstract | CrossRef Full Text | Google Scholar
19. Chen Y, Liu F, Nong Y, Ruan J, Guo Q, Luo 1000, et al. Clinical efficacy and machinery of growth hormone action in patients experiencing repeat implantation failure. Tin can J Physiol Pharmacol. (2018) 96:929–32. doi: 10.1139/cjpp-2017-0786
PubMed Abstruse | CrossRef Total Text | Google Scholar
20. Altmäe Southward, Mendoza-Tesarik R, Mendoza C, Mendoza N, Cucinelli F, Tesarik J. Effect of growth hormone on uterine receptivity in women with repeated implantation failure in an oocyte donation program: a randomized controlled trial. J Endocr Soc. (2018) two:96–105. doi: 10.1210/js.2017-00359
PubMed Abstract | CrossRef Full Text | Google Scholar
21. Cui N, Li AM, Luo ZY, Zhao ZM, Xu YM, Zhang J, et al. Effects of growth hormone on pregnancy rates of patients with sparse endometrium. J Endocrinol Invest. (2019) 42:27–35. doi: 10.1007/s40618-018-0877-i
CrossRef Full Text | Google Scholar
22. Yang J-Y, Li H, Lu N, Li L, Sun 10-X. Influence of growth hormone supplementation in patients with sparse endometrium undergoing frozen embryo transfer. Reprod Dev Med. (2019) 3:49–53. doi: 10.4103/2096-2924.255983
CrossRef Total Text | Google Scholar
23. Xue-Mei W, Hong J, Wen-Xiang Z, Yang 50. The furnishings of growth hormone on clinical outcomes later frozen – thawed embryo transfer. Int J Gynecol Obstet. (2016) 133:347–fifty. doi: 10.1016/j.ijgo.2015.x.020
PubMed Abstract | CrossRef Full Text | Google Scholar
24. Liu 10, Bai H, Xie J, Shi J. Growth hormone co-treatment on controlled ovarian stimulation in normal ovarian response women tin improve embryo quality. Gynecol Endocrinol. (2019) 35:787–91. doi: ten.1080/09513590.2019.1590545
PubMed Abstract | CrossRef Full Text | Google Scholar
25. Tesarik J, Hazout A, Mendoza C. Improvement of delivery and alive nativity rates after ICSI in women aged >40 years by ovarian co-stimulation with growth hormone. Hum Reprod. (2005) 20:2536–41. doi: 10.1093/humrep/dei066
PubMed Abstruse | CrossRef Full Text | Google Scholar
26. Yovich JL, Stanger JD. Growth hormone supplementation improves implantation and pregnancy productivity rates for poor-prognosis patients undertaking IVF. Reprod Biomed Online. (2010) 21:37–49. doi: x.1016/j.rbmo.2010.03.013
PubMed Abstract | CrossRef Total Text | Google Scholar
27. Duffy JM, Ahmad G, Mohiyiddeen 50, Nardo LG, Watson A. Growth hormone for in vitro fertilization. Cochrane Database Syst Rev. (2010) 7: CD000099. doi: ten.1002/14651858.CD000099.pub3
CrossRef Full Text | Google Scholar
28. Sbracia M, Scarpellini F, Poverini R, Alò PL, Rossi G, Di Tondo U, et al. Immunohistochemical localization of the growth hormone in human endometrium and decidua. Am J Reprod Immunol. (2004) 51:112–half-dozen. doi: 10.1046/j.8755-8920.2003.00127.x
PubMed Abstract | CrossRef Full Text | Google Scholar
29. Shifren JL, Osathanondh R, Yeh J. Human fetal ovaries and uteri: developmental expression of genes encoding the insulin, insulin-like growth factor I, and insulin-like growth factor II receptors. Fertil Steril. (1993) 59:1036–40. doi: ten.1016/S0015-0282(xvi)55924-X
PubMed Abstruse | CrossRef Full Text | Google Scholar
30. Thiet MP, Osathanondh R, Yeh J. Localization and timing of appearance of insulin, insulin-like growth cistron-I, and their receptors in the human fetal müllerian tract. Am J Obstet Gynecol. (1994) 170:152–half-dozen. doi: x.1016/S0002-9378(xiii)70297-vii
PubMed Abstract | CrossRef Full Text | Google Scholar
31. Lobie PE, Breipohl W, García Aragón J, Watesr MJ. Cellular localization of the growth hormone receptor/ binding protein in the male and female reproductive systems. Endocrinology. (1990) 126:2214–21. doi: 10.1210/endo-126-4-2214
PubMed Abstract | CrossRef Full Text | Google Scholar
32. Ferrara Northward. Vascular endothelial growth factor and the regulation of angiogenesis. Recent Prog Horm Res. (2000) 55:15–35.
PubMed Abstract | Google Scholar
33. Hannan NJ, Paiva P, Meehan KL, Rombauts LJF, Gardner DK, Salamonsen LA. Analysis of fertility-related soluble mediators in human uterine fluid identifies VEGF as a cardinal regulator of embryo implantation. Endocrinology. (2011) 152:4948–56. doi: ten.1210/en.2011-1248
PubMed Abstract | CrossRef Full Text | Google Scholar
34. Kaneko Y, Twenty-four hour period ML, White potato CR. Integrin β3 in rat blastocysts and epithelial cells is essential for implantation in vitro: studies with Ishikawa cells and small interfering RNA transfection. Hum Reprod. (2011) 26:1665–74. doi: 10.1093/humrep/der128
PubMed Abstract | CrossRef Full Text | Google Scholar
36. Tei C, Maruyama T, Kuji Northward, Miyazaki T, Mikami M, Yoshimura Y. Reduced expression of alphavbeta3 integrin in the endometrium of unexplained infertility patients with recurrent IVF-ET failures: improvement by danazol treatment. J Assist Reprod Genet. (2003) twenty:13–twenty. doi: 10.1023/A:1021254620888
PubMed Abstruse | CrossRef Full Text | Google Scholar
37. Juengel JL, Nett TM, Anthony R 5, Niswender GD. Effects of luteotrophic and luteolytic hormones on expression of mRNA encoding insulin-like growth gene I and growth hormone receptor in the ovine corpus luteum. J Reprod Fertil. (1997) 110:291–8. doi: x.1530/jrf.0.1100291
PubMed Abstruse | CrossRef Full Text | Google Scholar
38. Schams D, Berisha B, Kosmann 1000, Einspanier R, Amselgruber WM. Possible role of growth hormone, IGFs, and IGF-binding proteins in the regulation of ovarian office in large subcontract animals. Domest Anim Endocrinol. (1999) 17:279–85. doi: x.1016/S0739-7240(99)00044-2
PubMed Abstract | CrossRef Full Text | Google Scholar
40. Lanzone A, Di Simone N, Castellani R, Fulghesu AM, Caruso A, Mancuso S. Man growth hormone enhances progesterone production by human luteal cells in vitro: prove of a synergistic consequence with human chorionic gonadotropin. Fertil Steril. (1992) 57:92–half dozen. doi: 10.1016/S0015-0282(16)54782-7
PubMed Abstract | CrossRef Total Text | Google Scholar
41. Devoto L, Kohen P, Castro O, Vega M, Troncoso JL, Charreau East. Multihormonal regulation of progesterone synthesis in cultured homo midluteal cells. J Clin Endocrinol Metab. (1995) 80:1566–lxx. doi: ten.1210/jcem.80.v.7745001
PubMed Abstruse | CrossRef Total Text | Google Scholar
42. Taketani T, Yamagata Y, Takasaki A, Matsuoka A, Tamura H, Sugino N. Effects of growth hormone and insulin-similar growth factor 1 on progesterone production in man luteinized granulosa cells. Fertil Steril. (2008) 90:744–eight. doi: ten.1016/j.fertnstert.2007.07.1304
PubMed Abstract | CrossRef Full Text | Google Scholar
43. Wu H, Li 50, Li Chiliad, Yuan H. Effect of growth hormone on the endometrial and endometrial blood flow in forzen thawed embryo tranfer. J Reprod Med. (2013) 12:914–917.
Google Scholar
45. Conway-Campbell BL, Wooh JW, Brooks AJ, Gordon D, Brownish RJ, Lichanska AM, et al. Nuclear targeting of the growth hormone receptor results in dysregulation of cell proliferation and tumorigenesis. Proc Natl Acad Sci The states. (2007) 104:13331–6. doi: x.1073/pnas.0600181104
PubMed Abstract | CrossRef Full Text | Google Scholar
46. Homburg R, Eshel A, Abdalla HI, Jacobs HS. Growth hormone facilitates ovulation induction past gonadotrophins. Clin Endocrinol (Oxf). (1988) 29:113–7. doi: 10.1111/j.1365-2265.1988.tb00252.x
PubMed Abstract | CrossRef Full Text | Google Scholar
47. Owen EJ, Shoham Z, Mason BA, Ostergaard H, Jacobs HS. Cotreatment with growth hormone, afterward pituitary suppression, for ovarian stimulation in in vitro fertilization: a randomized, double-blind, placebo-control trial. Fertil Steril. (1991) 56:1104–10. doi: 10.1016/S0015-0282(16)54724-4
PubMed Abstract | CrossRef Full Text | Google Scholar
48. Homburg R, Singh A, Bhide P, Shah A, Gudi A. The re-growth of growth hormone in fertility handling: a critical review. Hum Fertil. (2012) fifteen:190–3. doi: 10.3109/14647273.2012.744855
PubMed Abstruse | CrossRef Full Text | Google Scholar
49. Eftekhar Thousand, Aflatoonian A, Mohammadian F, Eftekhar T. Adjuvant growth hormone therapy in antagonist protocol in poor responders undergoing assisted reproductive technology. Arch Gynecol Obstet. (2013) 287:1017–21. doi: 10.1007/s00404-012-2655-1
PubMed Abstract | CrossRef Total Text | Google Scholar
50. Bayoumi YA, Dakhly DMR, Bassiouny YA, Hashish NM. Addition of growth hormone to the microflare stimulation protocol among women with poor ovarian response. Int J Gynecol Obstet. (2015) 131:305–eight. doi: 10.1016/j.ijgo.2015.05.034
PubMed Abstruse | CrossRef Full Text | Google Scholar
51. Dakhly DMR, Bayoumi YA, Gad Allah SH. Which is the best IVF/ICSI protocol to be used in poor responders receiving growth hormone equally an adjuvant handling? A prospective randomized trial. Gynecol Endocrinol. (2016) 32:116–9. doi: 10.3109/09513590.2015.1092136
PubMed Abstruse | CrossRef Total Text | Google Scholar
52. Choe SA, Kim MJ, Lee HJ, Kim J, Chang EM, Kim JW, et al. Increased proportion of mature oocytes with sustained-release growth hormone handling in poor responders: a prospective randomized controlled study. Curvation Gynecol Obstet. (2018) 297:791–6. doi: 10.1007/s00404-017-4613-4
PubMed Abstract | CrossRef Total Text | Google Scholar
53. Dakhly DMR, Bassiouny YA, Bayoumi YA, Hassan MA, Gouda HM, Hassan AA. The addition of growth hormone adjuvant therapy to the long downward regulation protocol in poor responders undergoing in vitro fertilization: Randomized control trial. Eur J Obstet Gynecol Reprod Biol. (2018) 228:161–5. doi: 10.1016/j.ejogrb.2018.06.035
PubMed Abstract | CrossRef Full Text | Google Scholar
54. Yu H, Gao S, Tang H, Chen H, Deng Z, Yang L, et al. Growth hormone intrauterine perfusion combined with replacement cycle in the treatment of not-response sparse endometrium: report of 5 cases. Int J Clin Exp Med. (2016) ix:11982–9.
Google Scholar
55. Drakopoulos P, Pluchino N, Bischof P, Cantero P, Meyer P, Chardonnens D. Effect of growth hormone on endometrial thickness and fertility consequence in the handling of women with panhypopituitarism: a example report. J Reprod Med. (2016) 61:78–82.
PubMed Abstract | Google Scholar
56. Sebastian-León P, Garrido N, Remohí J, Pellicer A, Diaz-Gimeno P. Asynchronous and pathological windows of implantation: Two causes of recurrent implantation failure. Hum Reprod. (2018) 33:626–35. doi: 10.1093/humrep/dey023
PubMed Abstract | CrossRef Full Text | Google Scholar
57. Koot YEM, van SR, Boomsma CM, van Leenen D, Groot Koerkamp MJA, Goddijn 1000, et al. An endometrial gene expression signature accurately predicts recurrent implantation failure subsequently IVF. Sci Rep. (2016) 6:19411. doi: 10.1038/srep19411
PubMed Abstruse | CrossRef Full Text | Google Scholar
58. Altmäe S, Martinez-Conejero JA, Salumets A, Simon C, Horcajadas JAA, Stavreus-Evers A, et al. Endometrial gene expression analysis at the time of embryo implantation in women with unexplained infertility. Mol Hum Reprod. (2010) xvi:178–87. doi: x.1093/molehr/gap102
PubMed Abstruse | CrossRef Full Text | Google Scholar
59. Richter KS, Bugge KR, Bromer JG, Levy MJ. Human relationship betwixt endometrial thickness and embryo implantation, based on 1,294 cycles of in vitro fertilization with transfer of 2 blastocyst-stage embryos. Fertil Steril. (2007) 87:53–9. doi: ten.1016/j.fertnstert.2006.05.064
PubMed Abstract | CrossRef Full Text | Google Scholar
threescore. Abdalla HI, Brooks AA, Johnson MR, Kirkland A, Thomas A, Studd JWW. Endometrial thickness: a predictor of implantation in ovum recipients? Hum Reprod. (1994) 9:363–5. doi: ten.1093/oxfordjournals.humrep.a138509
PubMed Abstract | CrossRef Full Text | Google Scholar
61. Kasius A, Smit JG, Torrance HL, Eijkemans MJC, Mol BW, Opmeer BC, et al. Endometrial thickness and pregnancy rates later IVF: a systematic review and meta-analysis. Hum Reprod Update. (2014) xx:530–41. doi: 10.1093/humupd/dmu011
PubMed Abstract | CrossRef Full Text | Google Scholar
62. Miwa I, Tamura H, Takasaki A, Yamagata Y, Shimamura K, Sugino Due north. Pathophysiologic features of 'thin' endometrium. Fertil Steril. (2009) 91:998–1004. doi: 10.1016/j.fertnstert.2008.01.029
PubMed Abstract | CrossRef Full Text | Google Scholar
63. Chien L-Westward, Au H-K, Chen P-L, Xiao J, Tzeng C-R. Assessment of uterine receptivity past the endometrial-subendometrial blood flow distribution pattern in women undergoing in vitro fertilization-embryo transfer. Fertil Steril. (2002) 78:245–51. doi: 10.1016/S0015-0282(02)03223-5
PubMed Abstract | CrossRef Full Text | Google Scholar
65. Jiang H, Pei H, Zhang W, Wang X. A prospective clinical written report of interventional ultrasound sclerotherapy on women with hydrosalpinx before in vitro fertilization and embryo transfer. Fertil Steril. (2010) 94:2854–half dozen. doi: 10.1016/j.fertnstert.2010.06.065
PubMed Abstruse | CrossRef Full Text | Google Scholar
67. Altmäe S, Esteban FJ, Stavreus-Evers A, Simón C, Giudice L, Lessey BA, et al. Guidelines for the design, analysis and estimation of "omics" information: focus on human endometrium. Hum Reprod Update. (2014) xx:12–28. doi: 10.1093/humupd/dmt048
PubMed Abstruse | CrossRef Full Text | Google Scholar
68. Keane KN, Yovich JL, Hamidi A, Hinchliffe PM, Dhaliwal SS. Single-centre retrospective analysis of growth hormone supplementation in IVF patients classified as poor-prognosis. BMJ Open. (2017) 7:e018107. doi: ten.1136/bmjopen-2017-018107
PubMed Abstract | CrossRef Total Text | Google Scholar
Source: https://www.frontiersin.org/articles/10.3389/fendo.2019.00653/full
0 Response to "Artlabeling Activity Implantation of the Blastocyst in the Uterine Lining"
Post a Comment